Photo credit: Photos.com/Rice University via EurekAlert.org
Three patients with hereditary blood diseases have been cured by a new gene therapy with CRISPR-Cas, where doctors modified the DNA of red blood cell precursors. Scientists presented these results in June.
The development of the DNA editing method CRISPR-Cas created a revolution in micro and molecular biology, as this tool can accurately switch genes on and off. Researchers can also use this method to prevent certain diseases such as AIDS by repairing or modifying genes in embryos, like the Chinese scientist Jiankui He from Shenzhen University did with the twins Lulu and Nana.
Recently, American and Swiss scientists of the companies CRISPR Therapeutics and Vertex Pharmaceuticals Incorporated succeeded to cure diseases in adult patients with CRISPR-Cas for the first time. As a result, two patients with thalassemia and one patient with sickle cell disease no longer require medicine or blood transfusions.
Fetal hemoglobin
Sickle cell disease and thalassemia are hereditary forms of anemia. Both diseases are caused by an error in hemoglobin, a substance in the blood that transports oxygen around the body. Scientists activated an alternative form of hemoglobin with CRISPR-Cas, which is normally only active in babies in the womb: the fetal hemoglobin. That variant is error-free and can take over the role of the defective hemoglobin.
Within a baby’s first months, a transition takes place from fetal hemoglobin to the adult form of this oxygen transporter. At that time, a specific inhibitor blocks the production of the fetal hemoglobin. Gene therapy focuses on that inhibitor: the researchers cut the DNA coding for it with CRISPR-Cas. As a result, red blood cells once again contain fetal, and functional, hemoglobin. In the months following the gene therapy, the percentage of fetal hemoglobin in patients rose from almost zero to approximately fifty percent.
Outside the body
The gene therapy is carried out in the patient’s stem cells: the cells that produce all blood cells, including the red ones. Doctors isolated those stem cells from the patients’ bone marrow and genetically modified them in the laboratory. Stem cells that remain in the patient’s bone marrow, however, continue to produce red blood cells with the defective hemoglobin. Therefore, doctors killed these stem cells with chemotherapy before placing back the genetically modified stem cells.
Treating the stem cells outside the body is a great advantage according to John van der Oost, microbiologist at Wageningen University & Research in the Netherlands. He is not involved in the research but is a CRISPR-Cas pioneer. “Because the scientists make genetic adjustments in the laboratory, they can ‘read’ the DNA of the stem cells from start to finish after the CRISPR-Cas treatment and check whether the molecular tools have made the correct adjustments.” Doctors then only reintroduce those stem cells, whose DNA has the intended modifications, without additional changes to the DNA. “In this way, such gene therapy can be performed flawlessly,” says Van der Oost.
Bone pains
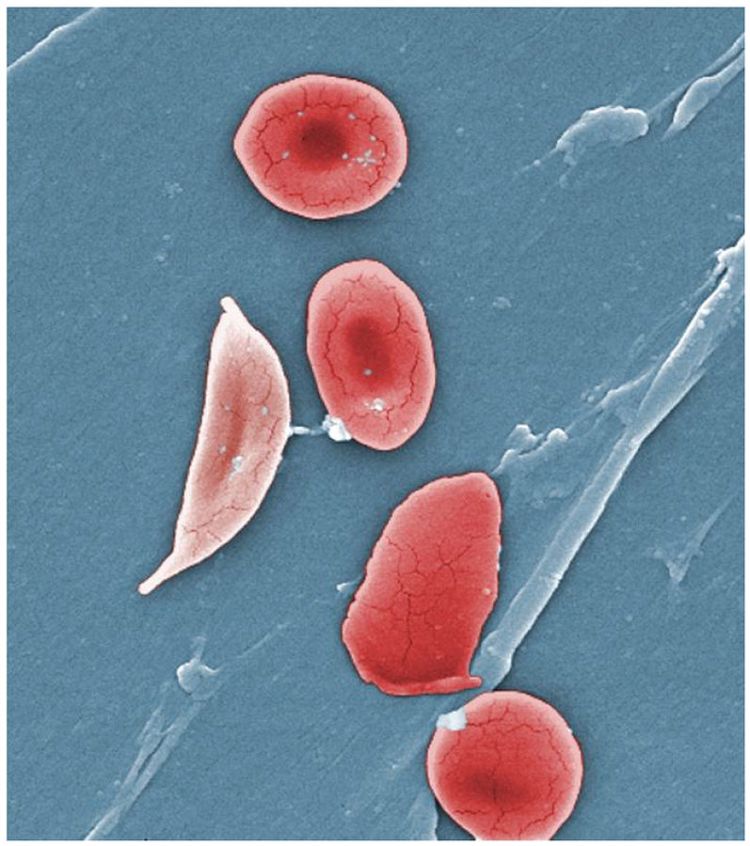
Every year, approximately 300,000 new sickle cell patients and 56,000 thalassemia patients are born worldwide. The disease mainly occurs in Africa, the Middle East and India, but has spread from there to the rest of the world. In both thalassemia and sickle cell disease, red blood cells live an average of thirty days, instead of the usual one hundred and twenty. This is because these red blood cells quickly break down due to their abnormal structure. In thalassemia patients, this sometimes happens even before the red blood cells leave the bone marrow. As a result, patients with severe thalassemia depend on blood transfusions throughout their lives. In sickle cell disease, the normally round red blood cells turn into sickle-shaped cells when they enter an oxygen-depleted environment. As a result, they stick together and to the blood vessel wall and clog small blood vessels. This happens throughout the body, but especially blockages in bones cause severe pains. In the long run, anemia, blockages (in sickle cell disease) and iron overload (in thalassemia) lead to organ damage. As a result, the average life expectancy for sickle cell and thalassemia patients lies between 50 and 55 years.
Stem cell transplants
Bart Biemond, internist-hematologist and professor of hemoglobinopathy at the Amsterdam UMC and the University of Amsterdam (UvA), is specialized in sickle cell disease and thalassemia. He is not involved in this specific research, but calls the therapy promising. “The patients in this study are cured of their condition and now have blood levels they never had before,” says Biemond. “I definitely see a future in this new gene therapy.”
Biemond and his colleagues from the Amsterdam UMC are currently treating sickle cell disease and thalassemia with stem cell transplantations. During this transplantation they replace the patient’s stem cells with those of a healthy sibling. “This method is successful: all transplants we have performed in adult sickle cell patients so far have been successful and it does not require chemotherapy,” says Biemond. Doctors perform stem cell transplantation in thalassemia almost exclusively in children, and for that treatment chemotherapy is necessary. The problem is that only one in ten patients has a suitable stem cell donor. A transplantation with genetically modified stem cells of the patient would therefore be a good alternative for those who do not have a suitable stem cell donor.
In addition to stem cell transplantations, doctors treat sickle cell disease with the ancient chemotherapeutic agent hydroxyurea. This drug, like the gene therapy, stimulates the production of fetal hemoglobin, but is less efficient. Other drugs prevent the sickle formation of the red blood cells or provide a higher oxygen uptake in the blood, but those drugs are not on the market yet. “An additional disadvantage is that these drugs do not prevent the severe bone pains patients experience,” says Biemond. Another medicine reduces the stickiness of the red blood cells and thus reduces the number of pain attacks, but does not cure the disease. Gene therapy does.
Heavy treatment
The clinical study of the CRISPR-Cas gene therapy for sickle cell disease and thalassemia is now in the initial phase, but Biemond expects this form of gene therapy to be on the market within five years. However, there are still some kinks to work out. “The principle sounds pretty simple, but it is a tough treatment,” Biemond warns. Patient receive high doses of chemotherapy to kill the untreated stem cells. Therefore, a patient must be fit enough to undergo gene therapy.
In addition, chemotherapy has side effects and it increases the risk of infections and could leads to infertility. Infertility is a problem, especially in bone marrow transplants or gene therapy in children, according to Biemond: “In adult patients, doctors can freeze sperm or egg cells, but in children those cells are not yet ripe enough.” That means that children who will be treated with this gene therapy in the future probably will not be able to have children themselves.
CRISPR-Cas therapy is not limited to hereditary blood disorders. Although it is currently difficult to repair a specific error in the DNA, doctors may also be able to use this technique in the future to cure other inherited disorders, such as muscular dystrophy.
Nicole van ‘t Wout Hofland wrote this article for biotechnologie.nl and NEMO Kennislink.
Reference
- Press release: CRISPR Therapeutics and Vertex Announce New Clinical Data for Investigational Gene-Editing Therapy CTX001™ in Severe Hemoglobinopathies at the 25th Annual European Hematology Association (EHA) Congress – CRISPR Therapeutics, 12 June 2020